InuMax® Advanced Retinol
* PRODUCT NAME:
• InuMax® Advanced Retinol
Next-generation encapsulated retinol – high efficacy, superior stability, and controlled irritation
* INCI NAME:
• Aqua/Water; Caprylic/Capric Triglyceride; Glycerin; Polysorbate 20; Retinol; Inulin Lauryl Carbamate; Behentrimonium Chloride; Sucrose Laurate; BHT; BHA; Phenoxyethanol; Benzoic Acid; Dehydroacetic Acid; Ethylhexylglycerin; Disodium EDTA
* PACKING:
• 1kg/5kg
* ORIGIN:
• Evonik-Germany
* APPEARANCE: pale yellow to yellow, slightly viscous liquid
1. OVERVIEW & TECHNOLOGICAL DIFFERENTIATION
• Retinol is a gold-standard anti-aging active ingredient but is well known for its limitations, including poor stability, high susceptibility to degradation, and a significant risk of skin irritation when used at higher concentrations.
• InuMax® Advanced Retinol is a next-generation retinol in which retinol is encapsulated in inulin using Evonik’s proprietary InuMax® technology. This encapsulation mechanism creates a fundamental difference in how retinol is delivered to and absorbed by the skin:
+ Free retinol: Rapid release → sudden high concentration of retinol on the skin surface → increased risk of stinging, redness, and irritation
+ InuMax® Advanced Retinol: Contains 8% encapsulated retinol within an inulin matrix (InuMax® technology), enabling a higher usable retinol load while minimizing skin irritation and significantly enhancing retinol stability.
Controlled and gradual release → more consistent penetration into the epidermal layers → reduced irritation risk while optimizing biological efficacy
• As a result, InuMax® Advanced Retinol enables higher retinol performance with improved skin tolerability.
2. SCIENTIFIC EVIDENCE
2.1. IN VITRO EFFICACY – CHEMICAL STABILITY
The chemical stability of retinol in InuMax® Advanced Retinol was monitored using HPLC and compared with non-encapsulated retinol at the same concentration.
• Storage conditions: Room temperature & 40°C
• Monitoring period: 28 weeks
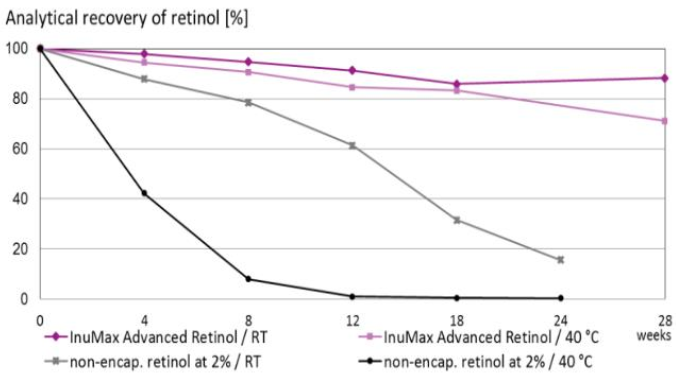
• 88% of retinol retained at room temperature after 28 weeks
• 72% of retinol retained at 40°C after 28 weeks
In contrast, a solution containing 2% free retinol showed:
• < 20% retinol remaining at room temperature
• < 1% retinol remaining at 40°C
→ Demonstrating the superior stability of the InuMax® encapsulation system
2.2 IN VIVO EFFICACY – BIOAVAILABILITY & SKIN PENETRATION
The penetration profile of InuMax® Advanced Retinol was evaluated using 5D-Intravital Tomography (5D-IVT) on human volunteers.
• Topical application of an aqueous solution containing 0.3% retinol
• Evaluation after 5 hours
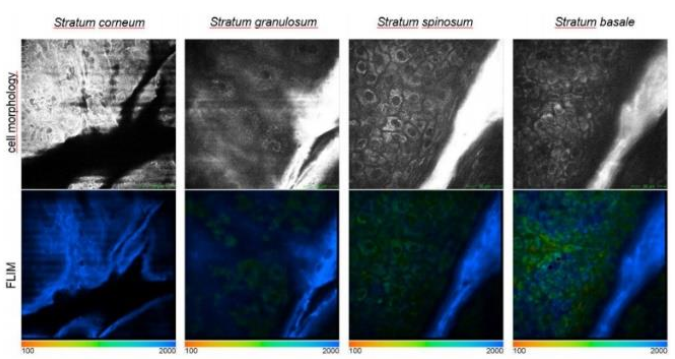
Observations:
• Retinol detected via FLIM fluorescence signal (blue)
• Penetration observed into the stratum spinosum and even reaching the stratum basale
→ Demonstrating good bioavailability and effective distribution of retinol into deeper epidermal layers
2.3 SKIN IRRITATION STUDY (IN VIVO)
A comparative irritation study was conducted on three volunteers to evaluate free retinol versus InuMax® Advanced Retinol.
• Test samples:
– Aqueous solution containing 0.3% free retinol
– Solution containing InuMax® Advanced Retinol, equivalent to 0.3% retinol
• Method: Occlusive application on the forearm
• Evaluation period: 48 hours
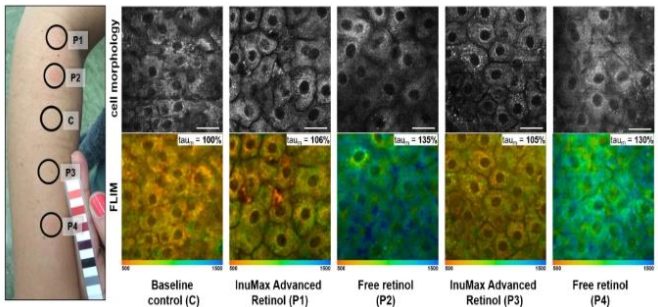
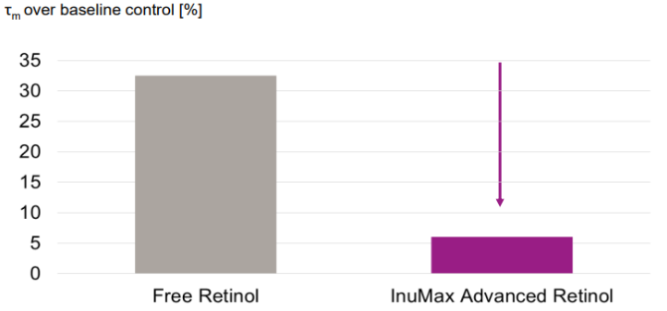
=> Cellular inflammatory activity was assessed via NADH fluorescence lifetime (Tauₘ), a marker reflecting cellular metabolic stress.
Results:
• Skin treated with free retinol showed significantly higher Tauₘ values
• Free retinol increased Tauₘ by approximately 33% compared to the control
• InuMax® Advanced Retinol caused only a minimal increase in Tauₘ, indicating markedly lower irritation
→ Confirming the advantage of controlled-release technology in reducing cellular stress and inflammatory responses in keratinocytes












